 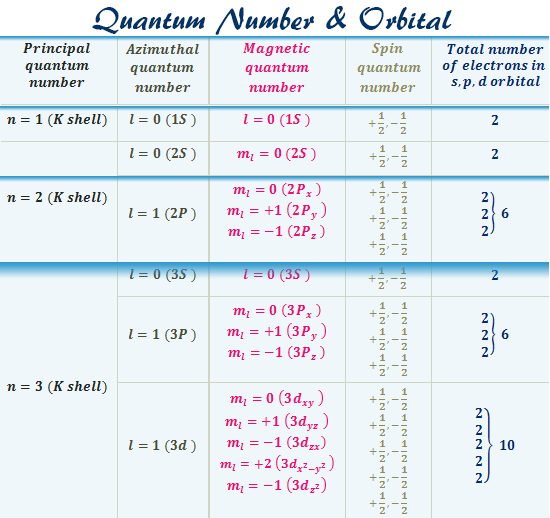 
If electronegativity values aren't given, you should assume that a covalent bond is polar unless it is between two atoms of the same element. So in general, we can predict that any metal-nonmetal combination will be ionic and any nonmetal-nonmetal combination will be covalent. No two electrons in the same atom can have the same set of values for all the four quantum numbers. In addition, each electron has a spin quantum number, ms, that can be equal to 12. The number of angular (planar) nodes in an orbital is Np l. n1 This quantum number labels subshells (and orbitals therein) and determines the general shape of the orbitals within the subshell. Number of Radial nodesnl1 Number of angularnodesl Where lAzimuthalquantumnumbernprincipalquantumnumber For 4d orbital n4 andl2 In4d orbital the number. Metals have low electronegativity compared to nonmetals. The magnetic quantum number, ml, with 2l + 1 values ranging from l to +l, describes the orientation of the orbital in space. The energy of orbitals in shell n is En, where k 2.179x1018 J 2 Angular Momentum Quantum Numberl 0, 1, 2, 3. The first three ( n, l, ml) specify the particular orbital of interest, and the fourth ( ms) specifies how many electrons can occupy that orbital. Hence, this set represents electrons in a 4d orbital. Each electron in an atom is described by four different quantum numbers. If you are not given electronegativity values, you can still predict the bond type using the periodic table. In the set of quantum numbers 4, 2, 1, -1/2, n 4, l 2, ml 1, s -1/2, l 2 is for d orbital. If the difference is between 0.4 and 2.0 units, the bond is classified as polar covalent, and if the difference is more than 2.0 units, the bond is substantially ionic. If the electronegativities differ by less than 0.4 units, the bond can be classified as nonpolar covalent. One method to classify bonds based on this difference can be described as follows. Electronegativity difference can be used to predict bond type. An ionic bond results when the sharing is so unequal that fully charged ions form. The magnetic quantum number, ml, tells you exactly in which orbital you can expect to find the electron. (ii) Azimuthal or angular momentum quantum number (l. It also tells the maximum number of electrons a shell can accommodate is 2n 2, where n is the principal quantum number. A polar covalent bond is one in which the electrons are unequally shared between the atoms. (i) Principal quantum number (n): It tells the main shell in which the electron resides and the approximate distance of the electron from the nucleus. The letter in the orbital name defines the subshell with a specific angular momentum quantum number l 0 for s orbitals, 1 for p orbitals, 2 for d orbitals. When two bonded atoms attract electrons with equal strength, the result is a nonpolar covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
